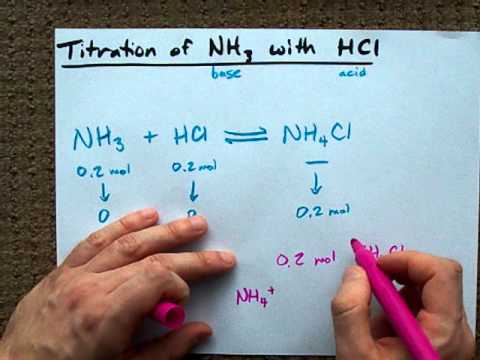
![Prodibio - Prodibio Start Up Nano [4 ampułki] - ogranicza wzrost niejonowego amoniaku NH3 i produkcję azotynów - sklep akwarystyczny Prodibio - Prodibio Start Up Nano [4 ampułki] - ogranicza wzrost niejonowego amoniaku NH3 i produkcję azotynów - sklep akwarystyczny](https://sklep.roslinyakwariowe.pl/zdjecia/prodibio_start_up_nano_4_ampulki_ogranicza_wzrost_niejonowego_amoniaku_nh3_i_produkcje_azotynow-i-36266-1.jpg)
Prodibio - Prodibio Start Up Nano [4 ampułki] - ogranicza wzrost niejonowego amoniaku NH3 i produkcję azotynów - sklep akwarystyczny
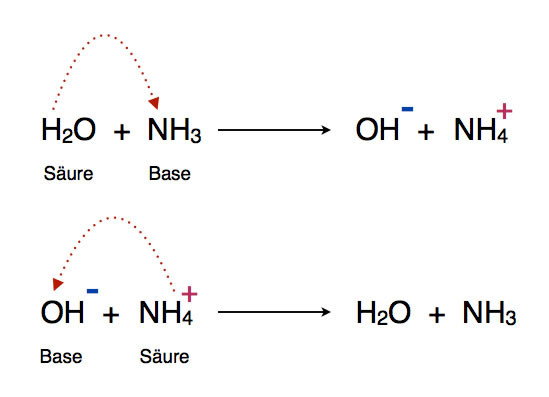
Welche Bedeutung hat die chemische Formel,,H2O+NH3- - - > OH- +NH4“ für die Säure-Base-Eigenschaften von Ammoniak? (Chemie, Säure-Base-Reaktion)
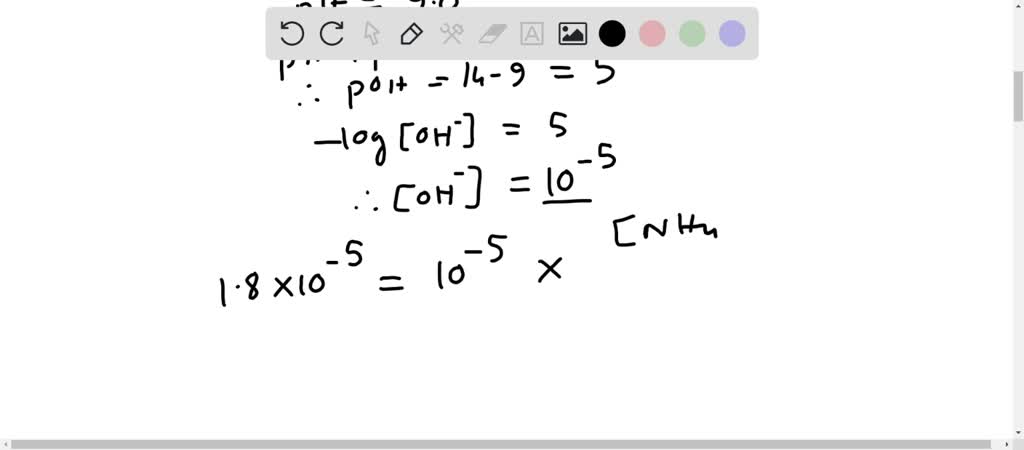
SOLVED: How many moles of NH4Cl must be added to 2.0 L of 0.10M NH3 to form a buffer whose pH is 9.00? (Assume that the addition of NH4Cl does not change

SOLVED: Which solution has the greatest buffering capacity?A) 0.335 M NH3 and 0.100 M NH4ClB) 0.085 M NH3 and 0.090 M NH4ClC) 0.540 M NH, and 0.550 M NH4ClD) 0.200 M NH,